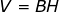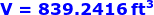Category: Chemical Engineering Math
"Published in Newark, California, USA"
The following reaction
was allowed to proceed to equilibrium. The contents of the two-liter reaction vessel were then subjected to analysis and found to contain 1.0 mole H2S, 0.20 mole H2, and 0.80 mole S2. What is the equilibrium constant Keq for this reaction?
Solution:
Since the amount of H2S, H2, and S2 are given already in moles and they are in a 2L reaction vessel, then we can calculate their concentration in molarity.
Molarity of H2S:
Molarity of H2:
Molarity of S2:
Consider the given reaction
The equilibrium constant for the reaction is
The coefficient of each substance in the reaction is equal to the exponent of each factor for the equation above. The units of concentration of substances are always in molarity. Therefore, the equilibrium constant for the reaction is
Note: Usually, the value of Keq is unitless because the units of the concentration of substances are not included in the substitution to the equation of chemical equilibrium. If Keq = 0, then no reaction nor conversion of reactants to products involved in the process and on the other hand, if Keq = ∞, then the reaction is 100% complete in the process.

This website will show the principles of solving Math problems in Arithmetic, Algebra, Plane Geometry, Solid Geometry, Analytic Geometry, Trigonometry, Differential Calculus, Integral Calculus, Statistics, Differential Equations, Physics, Mechanics, Strength of Materials, and Chemical Engineering Math that we are using anywhere in everyday life. This website is also about the derivation of common formulas and equations. (Founded on September 28, 2012 in Newark, California, USA)
Friday, September 5, 2014
Thursday, September 4, 2014
Limiting and Excess Reagent Problems
Category: Chemical Engineering Math
"Published in Newark, California, USA"
What is the maximum weight of SO3 that could be made from 25.0 grams of SO2 and 6.0 grams of O2 by the following reaction? (Atomic Weights: S = 32 and O = 16)
Solution:
The first thing that we need to do is to get the molecular weights of SO2, O2, and SO3 from the given atomic weights.
Molecular Weight of SO2:
Molecular Weight of O2:
Molecular Weight of SO3:
Next, we will calculate the amount of SO3 which is the product of SO2 and O2. From the given reaction
Weight of SO3 from SO2:
Weight of SO3 from O2:
From the two reactants, O2 will give us the least amount of product which is SO3. O2 is the limiting reagent. On the other hand, SO2 is the excess reagent. Once O2 is completely converted into SO3, there will be a left over or unused amount of SO2 in the reaction.
In this problem, we have to choose the limiting reagent to determine the amount of product produced. Therefore, the maximum weight of SO3 is
"Published in Newark, California, USA"
What is the maximum weight of SO3 that could be made from 25.0 grams of SO2 and 6.0 grams of O2 by the following reaction? (Atomic Weights: S = 32 and O = 16)
Solution:
The first thing that we need to do is to get the molecular weights of SO2, O2, and SO3 from the given atomic weights.
Molecular Weight of SO2:
Molecular Weight of O2:
Molecular Weight of SO3:
Next, we will calculate the amount of SO3 which is the product of SO2 and O2. From the given reaction
Weight of SO3 from SO2:
Weight of SO3 from O2:
From the two reactants, O2 will give us the least amount of product which is SO3. O2 is the limiting reagent. On the other hand, SO2 is the excess reagent. Once O2 is completely converted into SO3, there will be a left over or unused amount of SO2 in the reaction.
In this problem, we have to choose the limiting reagent to determine the amount of product produced. Therefore, the maximum weight of SO3 is
Wednesday, September 3, 2014
Mole Fraction Problems
Category: Chemical Engineering Math
"Published in Newark, California, USA"
Calculate the mole fractions of ethyl alcohol, C2H5OH, and water in a solution made by dissolving 9.2 grams of alcohol in 18 grams of H2O. (Atomic Weights: H = 1, C = 12, and O = 16)
Solution:
There are two substances or components in a solution which are ethyl alcohol (solute) and water (solvent). The first thing that we need to do is to get the molecular weight of each component.
Molecular Weight of Ethyl Alcohol:
Molecular Weight of Water:
Next, we can get the moles of each component since we know their molecular weights.
Moles of Ethyl Alcohol:
Moles of Water:
Therefore, the mole fraction of ethyl alcohol is
and the mole fraction of water is
Note: Mole fractions are unitless and the sum of the mole fractions of components in a solution must be equal to 1.
"Published in Newark, California, USA"
Calculate the mole fractions of ethyl alcohol, C2H5OH, and water in a solution made by dissolving 9.2 grams of alcohol in 18 grams of H2O. (Atomic Weights: H = 1, C = 12, and O = 16)
Solution:
There are two substances or components in a solution which are ethyl alcohol (solute) and water (solvent). The first thing that we need to do is to get the molecular weight of each component.
Molecular Weight of Ethyl Alcohol:
Molecular Weight of Water:
Next, we can get the moles of each component since we know their molecular weights.
Moles of Ethyl Alcohol:
Moles of Water:
Therefore, the mole fraction of ethyl alcohol is
and the mole fraction of water is
Note: Mole fractions are unitless and the sum of the mole fractions of components in a solution must be equal to 1.
Tuesday, September 2, 2014
More Cylinder Problems, 13
Category: Solid Geometry
"Published in Vacaville, California, USA"
The outer protective smokestack of a steamship is streamlined so that it has a uniform oval section parallel to the deck. The area of this oval section is 48 sq. ft. If the length of the stack is 18 ft. and if the axis is raked 76°15' to the horizontal, find the volume enclosed by the stack. (Raked means inclined.)
Solution:
To illustrate the problem, it is better to draw the figure as follows
The area of a base which is an oval section is already given in the problem. By using a trigonometric function of a right triangle, the altitude of the smokestack is
Therefore, the volume of a smokestack which is a cylinder is
"Published in Vacaville, California, USA"
The outer protective smokestack of a steamship is streamlined so that it has a uniform oval section parallel to the deck. The area of this oval section is 48 sq. ft. If the length of the stack is 18 ft. and if the axis is raked 76°15' to the horizontal, find the volume enclosed by the stack. (Raked means inclined.)
Solution:
To illustrate the problem, it is better to draw the figure as follows
 |
| Photo by Math Principles in Everyday Life |
The area of a base which is an oval section is already given in the problem. By using a trigonometric function of a right triangle, the altitude of the smokestack is
Therefore, the volume of a smokestack which is a cylinder is
Subscribe to:
Posts (Atom)