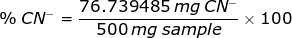Category: Chemical Engineering Math
"Published in Newark, California, USA"
An EDTA solution prepared from its disodium salt was standardized using 506.3 mg of primary standard CaCO3 and consumed 28.50 mL of the solution. The standard solution was used to determine the hardness of a 2L sample of mineral water, which required 35.57 mL EDTA solution. Express the analysis in terms of ppm CaCO3.
Solution:
If CaCO3 is used to standardized EDTA disodium salt solution, then the chemical reaction is
From the chemical reaction above, we can calculate the concentration of EDTA disodium salt solution as follows
If the standardized EDTA disodium salt solution is used to determine the hardness of mineral water, then the amount of CaCO3 is
Therefore, the amount of CaCO3 in ppm is
Category: Chemical Engineering Math
"Published in Newark, California, USA"
A 750.25 mg alloy of nickel was dissolved and treated to remove the impurities. The ammonium solution was treated with 50 mL of 0.1075 M KCN and the excess cyanide required 2.25 mL of 0.00925 M AgNO3. Determine the % Ni in the alloy.
Solution:
If nickel is dissolved in ammonium solution, then nickel is converted into nickel ion in order to remove the impurities. If KCN solution is added to nickel solution, then the chemical reaction is
From the chemical reaction above, we can solve for the weight of nickel as follows
Therefore, the percent of nickel in the alloy is
Category: Chemical Engineering Math
"Published in Newark, California, USA"
A 500 mg sample containing NaCN required 23.50 mL of 0.1255 M AgNO3 to obtain a permanent faint turbidity. Express the result of this analysis as %CN-.
Solution:
If AgNO3 is added to NaCN, then the chemical reaction is
From the chemical reaction above, the weight of CN- is
Therefore, the percent of CN- in a sample is